Advancing medical device development with ex vivo human skin
Medical devices are evolving rapidly, becoming more sophisticated and diverse in their applications. From injectable drug delivery systems to wound dressings, wearable sensors, and microneedles, these innovations must undergo rigorous testing to ensure safety, functionality, and biocompatibility. However, as medical devices become more complex, so do the challenges associated with their preclinical evaluation, especially when it comes to predicting human skin responses accurately.
A new approach: ex vivo human skin models
HypoSkin®, Genoskin’s proprietary ex vivo human skin model, bridges this gap by preserving real, immunocompetent skin in a controlled environment. It enables more predictive, efficient, and ethical testing of medical devices, leading to faster development timelines and safer products.
In this blog, we will explore the challenges and limitations of traditional testing methods, how HypoSkin® is revolutionizing medical device testing, and highlight why it outperforms traditional models.
Scientific context: The field of medical devices
A medical device is any instrument, apparatus, or implant used to diagnose, prevent, or treat diseases without relying primarily on pharmacological action. This broad category includes injectable drug delivery systems, wearable sensors, wound dressings, microneedles, and adhesive patches, all of which require rigorous testing to ensure safety, efficacy, and regulatory compliance.
The development process for medical devices follows a structured workflow, starting with concept and design, followed by prototype development, preclinical testing, and ultimately clinical trials prior to acquiring regulatory approval. Early-stage testing typically involves in vitro studies to assess basic safety, followed by animal testing to evaluate biocompatibility and performance in a living system. If successful, devices move into human clinical trials, where regulatory agencies such as the FDA or EMA determine whether they meet safety and efficacy standards.
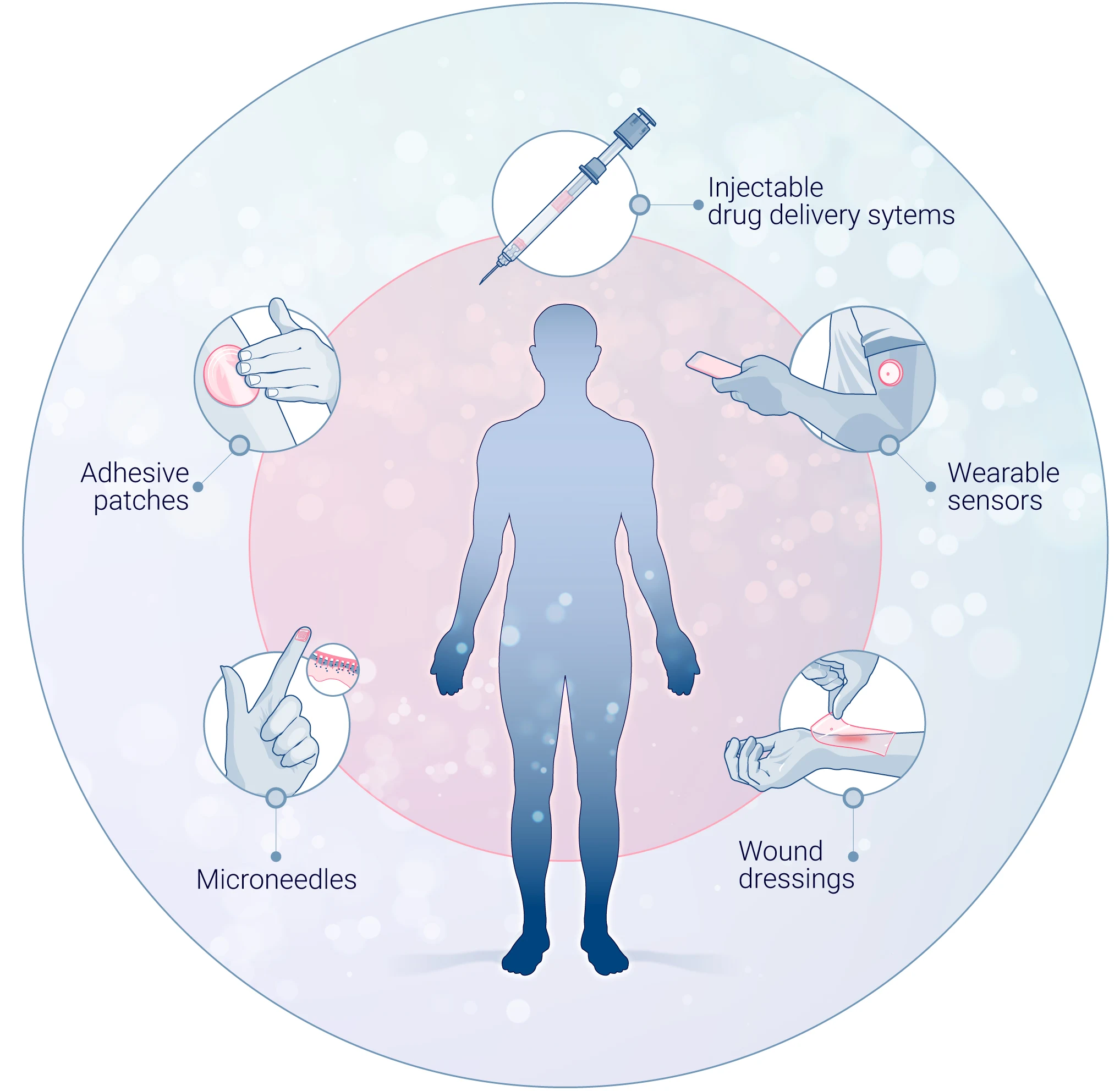
The growing need for reliable medical device testing
As medical devices become more advanced, regulatory agencies are imposing stricter safety and biocompatibility requirements, particularly for those that interact with the skin. Standards like ISO 10993-1 outline the need for rigorous biocompatibility assessments, while regulatory agencies demand more human-relevant data before approving new devices. This increasing regulatory scrutiny highlights the need for more predictive preclinical models to ensure safety and efficacy before clinical trials.
Traditional testing methods face significant limitations. In vitro models, while useful for preliminary assessments, lack the complexity of human skin, failing to replicate its barrier function, immune response, and structural integrity. Animal models, long used in medical device testing, pose ethical concerns and suffer from poor translational value—differences in skin composition, immune responses, and healing mechanisms often lead to misleading results. Clinical trials, though necessary, are expensive, time-consuming, and constrained by regulatory hurdles, making early detection of safety concerns critical to prevent costly failures.
Given these challenges, medical device developers must adopt more advanced, human-relevant testing strategies to meet regulatory demands, minimize risks, and accelerate market approval. Ex vivo human skin models like HypoSkin® provide a transformative solution, offering a highly predictive, ethical, and cost-effective alternative to traditional methods. By enabling realistic preclinical assessments, these models can help bridge the gap between device innovation and regulatory approval, ensuring safer products reach patients faster.
HypoSkin®: A game-changer for medical device testing
HypoSkin®, Genoskin’s injectable ex vivo human skin model, offers a transformative approach to evaluating biocompatibility, toxicity, and immune responses before entering clinical trials. Unlike traditional models, HypoSkin® maintains real, immunocompetent human skin alive, preserving its structural integrity and immune functions for up to 7 days in a controlled lab environment. This enables medical device developers to conduct highly predictive studies on how their products interact with real human tissue, providing a level of accuracy that in vitro and animal models cannot match.
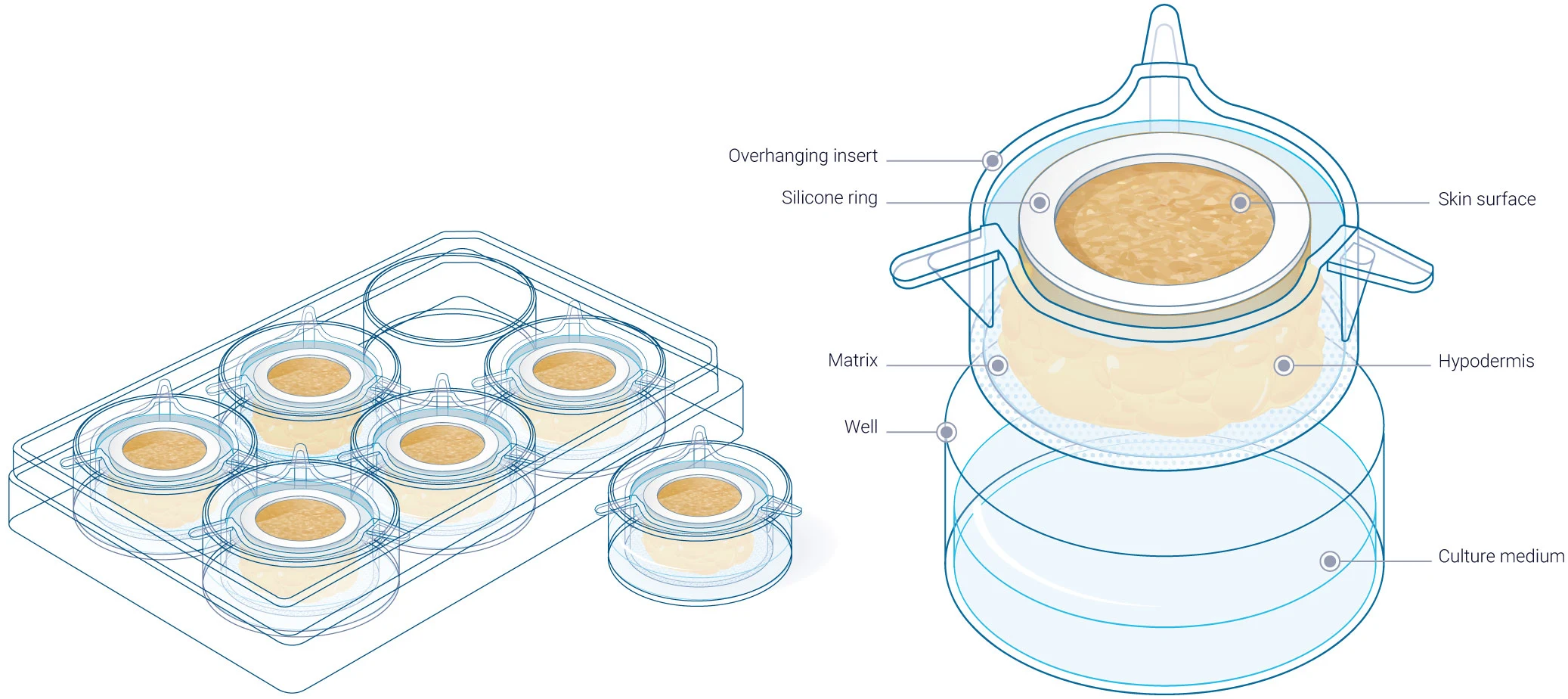
Why testing on real human skin matters?
Medical devices, particularly those that come into direct contact with the skin, must be assessed for their potential toxic, inflammatory, or sensitizing effects. HypoSkin® bridges the gap between traditional testing models and regulatory requirements by offering a realistic testing environment, allowing developers to:
- Mimic real-life conditions, ensuring the device interacts with human skin as it would in actual use.
- Assess immune responses, including inflammation and potential irritation caused by foreign materials, adhesives, or injected substances.
- Eliminate interspecies differences, ensuring results are directly translatable to human applications, reducing the risk of late-stage failures in clinical trials.
Use cases for medical device testing with HypoSkin®
1. Biocompatibility and Toxicity Testing
Ensuring that medical devices do not induce adverse reactions is a key regulatory requirement. HypoSkin® provides a human-relevant platform for biocompatibility testing, offering insights into potential toxicity, irritation, and inflammatory responses. In a study on macroencapsulation devices for therapeutic protein delivery1, researchers assessed the material’s biocompatibility by implanting an empty capsule into tissue. Results showed no major necrosis or fibrosis, and the material itself did not trigger pro-inflammatory cytokine release, confirming its safety.
2. Performance and Efficacy Testing
Testing medical devices in real human skin is crucial for ensuring they function as intended. HypoSkin® enables realistic evaluation of drug delivery systems, such as cell-based therapies. In a study using immortalized human myoblasts in encapsulated cell technology2, researchers demonstrated sustained protein release over time within an implanted skin model, as expected for this specific technology.
3. Inflammation and Immune Response Profiling
Understanding immune activation is essential for predicting inflammatory risks associated with medical devices. HypoSkin® preserves functional immune cells, allowing precise evaluation of immune responses. In a study on NIR-activated hydrogels for metformin delivery3, researchers found that light-triggered drug release affected local cytokine expression and immune pathways. HypoSkin® can similarly assess how materials or devices influence immune activation, helping to minimize adverse inflammatory reactions while ensuring therapeutic efficacy.
By offering a more predictive, ethical, and cost-effective solution, HypoSkin® is revolutionizing medical device testing, helping developers bring safer, more effective products to market faster.
Conclusion
The growing complexity of medical devices demands more predictive, ethical, and efficient testing solutions. Traditional models—whether in vitro, animal-based, or early clinical trials—often fall short of accurately predicting human responses, leading to costly failures and regulatory delays. HypoSkin® bridges this gap by providing an ex vivo human skin model that preserves the structural integrity and immune competence of real human skin. This enables developers to assess biocompatibility, performance, and immune response with unparalleled accuracy, reducing reliance on animal testing and expediting regulatory approval.
Looking ahead, ex vivo human skin models like HypoSkin® will play an increasingly critical role in personalized medicine and the development of next-generation medical devices. From customized drug delivery systems to advanced biomaterials and implantable technologies, these models will enable more precise, patient-specific testing, leading to safer and more effective innovations.
If you’re looking to enhance your medical device development with human-relevant testing, we invite you to explore HypoSkin®. Download our white paper or request a consultation with Genoskin experts to see how this innovative platform can accelerate your product development and regulatory approval process.
References
1. Engineering a versatile and retrievable cell macroencapsulation device for the delivery of therapeutic proteins
Grogg J, Vernet R, Charrier E, et al. iScience. 2023;26(8):107372. Published 2023 Jul 13. doi:10.1016/j.isci.2023.107372
2. Immortalized human myoblast cell lines for the delivery of therapeutic proteins using encapsulated cell technology
Lathuiliere, Aurelien et al. Molecular Therapy Methods & Clinical Development, Volume 26, 441 – 458, August 2022. DOI: 10.1016/j.omtm.2022.07.017
3. Near-infrared light activatable hydrogels for metformin delivery – Nanoscale (RSC Publishing)
Li Chengnan et al. Nanoscale, September 2019, Issue 34. https://doi.org/10.1039/C9NR02707F
Comments are closed.





