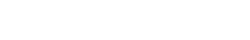Comprehensive ISR testing for injectable therapeutics

The ISR Platform® is a non-clinical service for Injection Site Reaction assessment in ex vivo human skin. Designed to support the development of injectable therapeutics and drug delivery systems, it evaluates local immune activation following subcutaneous and intradermal administration using a human-relevant New Approach Methodology (NAMs) to inform decision-making.
The ISR Platform® is built as a flexible service with configurable endpoints to answer your therapeutic specific requirements. Readouts can include secreted inflammatory mediators, gene expression signatures, and additional biomarker and histology-based assessments, providing human-relevant data to support development decisions.
Addressing Injection Site Reaction risk before clinical trials

Understanding and managing ISR risk in non-clinical development
Injection site reactions (ISR) are among the most frequently reported adverse events associated with subcutaneous and intradermal therapeutics (1,2). Local inflammation, erythema, swelling, induration, and injection site pain can directly impact patient compliance, dosing strategy, and product differentiation.
For biologics, advanced therapies, and complex drug delivery systems, ISR risk may arise from multiple sources—including the active compound, excipients, formulation characteristics, or device-related factors. Without early evaluation, these risks may only become apparent during clinical development, potentially leading to reformulation efforts, protocol adjustments, or program delays.
A targeted non-clinical assessment of local immune activation is therefore critical to support candidate selection, formulation optimization, and overall development strategy. The ISR Platform® provides human-relevant data in ex vivo human skin to help sponsors understand and manage ISR risk before clinical exposure.
1. Zhi, L., Liu, D. & Shameem, M. Injection site reactions of biologics and mitigation strategies. AAPS Open 11, 5 (2025). https://doi.org/10.1186/s41120-025-00108-4
2. Adam Felman. What to know about injection site reactions. Medical News Today July 16, 2025

Human-relevant mechanistic insight into Injection Site Reactions (ISR)
Understanding injection site reactions requires more than measuring isolated inflammatory markers. ISR involves coordinated local immune activation, cellular recruitment, and tissue-level responses following administration of an injectable product.
The ISR Platform® leverages injectable, immunocompetent ex vivo human skin to characterize local immune and tissue responses in a physiologically relevant environment. This approach enables assessment of:
- Local inflammatory mediator release
- Gene expression signatures associated with immune activation
- Tissue-level changes following administration
- Donor-to-donor variability in ISR response
Genoskin’s proprietary bioinformatics applications support the interpretation of complex, multi-omics datasets (including secretomic and transcriptomic data), helping sponsors extract detailed insights and inform key development decisions.
By combining human biology with configurable analytical endpoints, the ISR Platform® supports a deeper understanding of injection site reactions to guide development strategy.

Is it the right time to assess Injection Site Reaction risk?
Candidate selection
- Compare lead candidates for relative ISR liability
- Support down-selection using human-relevant local immune activation data
Formulation optimization
- Assess the impact of excipients, buffers and concentration changes
- Compare formulations to support optimization before clinical evaluation
Route strategy and lifecycle management
- Evaluate local tolerability for subcutaneous and intradermal administration
- Support IV-to-subcutaneous (SC) repurposing decisions
Device and delivery system development
- Assess ISR risk for drug delivery systems and combination products
- Evaluate administration parameters under clinically relevant conditions
De-risking before first-in-human
- Generate human-relevant data to support internal safety discussions
- Inform dose and administration strategy to guide development decisions
Human-relevant assessment of local immune and tissue responses following injectable administration

Bulk RNA sequencing reveals local immune activation pathways
Bulk RNA sequencing performed on treated ex vivo human skin enables comprehensive characterization of gene expression changes associated with injection site reactions. Transcriptomic profiling captures coordinated immune activation pathways, inflammatory signaling, and tissue response signatures following administration of injectable compounds.
It provides mechanistic insight into local immune activation and support comparative evaluation of candidates, formulations, and administration strategies within a human-relevant non-clinical framework.
Here, we present data published in August 2024 in Allergy. A mRNA Covid vaccine was injected subcutaneously into the HypoSkin® model; The RNA was collected at 8h and 24h post-injection.
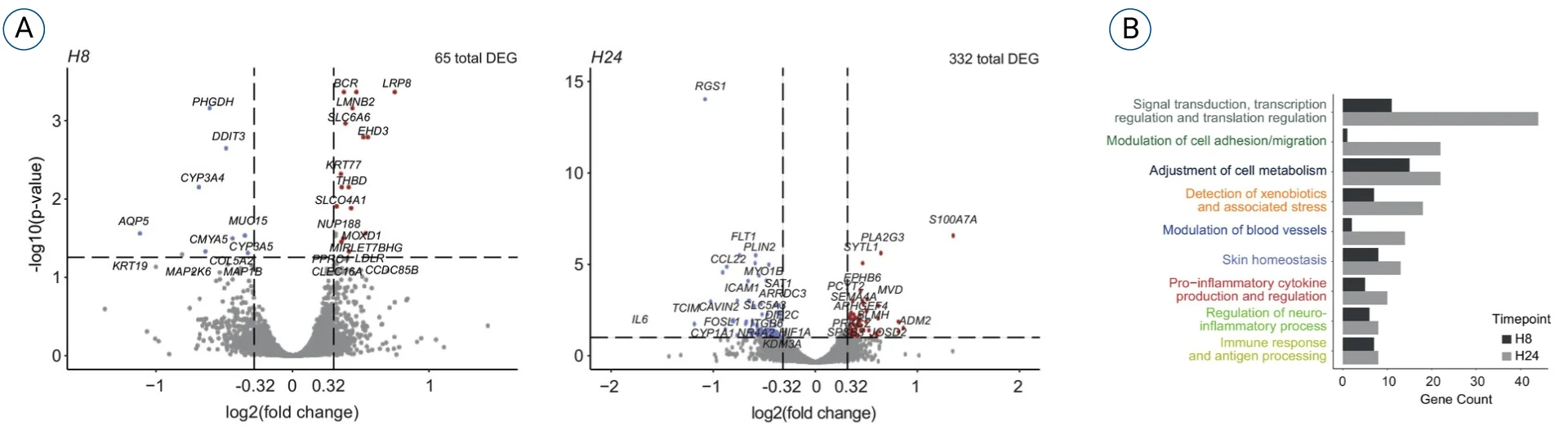
The figures show 9 different categories of pathways at 8h and 24h post injection.
While cell migration and antigen processing are expected in the context of vaccine exposure, biological pathways such as the detection of xenobiotics and stress, the neuroinflammation and the blood vessel modulation could be interpreted as signs of injection site and pain reactions.
Summary of results:
- Differential expression: 65 DEGs at 8h and 322 DEGs at 24h.
- Interpretation: Pathway-level analysis performed by our scientific team using FindYourPath.
- What we observe: Enrichment across 9 pathway categories, including:
- signal transduction / transcriptional regulation
- cell adhesion & migration
- immune response & antigen processing
- pro-inflammatory cytokine production & regulation
- neuro-inflammatory processes and blood vessel modulation
- Takeaway: Transcriptomic profiling captures a time-dependent escalation of local immune and tissue-response signatures consistent with ISR-related biology.
After subcutaneous or intradermal treatment of models at Day 1, culture medium are sampled to perform multiplex cytokine analysis.
The ISR Platform® service is recommanded with several donors in triplicates to ensure statistically significant results of cytokine expression. This allows for more accurate characterization of the activated biological pathways implicated in the local immune response.
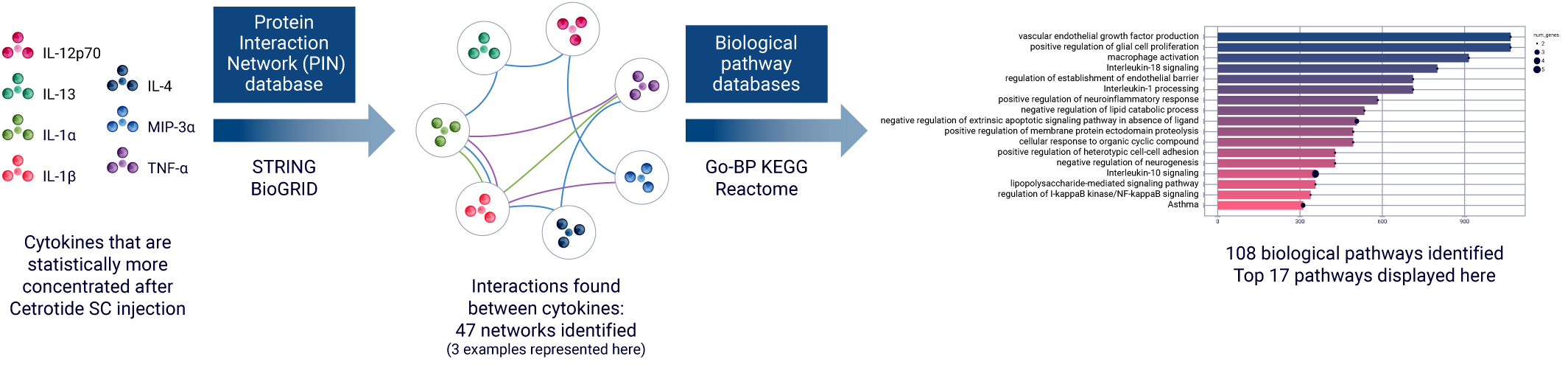
The exponential increase in biological data complexity has reached a point where even highly skilled experts find data analysis incredibly challenging. With this in mind, Genoskin created AUDACY (Automated Data Analysis of Cytokines), a proprietary bioinformatics-based analytical solution.
AUDACY is designed to:
- Manage large datasets and provide results quickly,
- Minimize the introduction of human error into the analysis,
- Enable novel forms of analysis.
Utilizing data from publicly available, peer reviewed online database such as PIN (Protein Interaction Network) or Reactome, AUDACY supports understanding interconnections between cytokines and gaining insight into the specific mechanisms that have been activated following the administration of the compound into the skin. Ultimately, we are able to infer on biological pathways implicated correlating the data generated ex vivo with clinically-related manifestations.
Bioinformatics-based analysis of multiplex cytokine assays with AUDACY
Frequently asked questions
How many donors are required to generate statistically significant data?
What is Injection Site Reaction (ISR) testing in ex vivo human skin?
Unlike simplified in vitro systems, ex vivo human skin preserves native immune cell populations and tissue architecture, providing a biologically relevant framework for evaluating local tolerability. ISR testing can support candidate selection, formulation optimization, and route strategy decisions within a controlled, translational setting.
ISR assessment may include:
- Measurement of inflammatory mediators
- Transcriptomic profiling
- Evaluation of donor variability
Why assess Injection Site Reactions (ISR) during non-clinical development?
Without early assessment, ISR liability may only become apparent during clinical studies, potentially requiring reformulation, route adjustment, or dose modification. A human-relevant, non-clinical ISR evaluation supports informed decisions around candidate selection, formulation design, route strategy, and overall development planning.
By integrating ISR testing early in development, sponsors can proactively manage local tolerability risk and strengthen the scientific rationale supporting clinical progression.
How can ISR testing support IV-to-subcutaneous (SC) repurposing?
Non-clinical ISR assessment in ex vivo human skin enables evaluation of local inflammatory and tissue responses under clinically relevant subcutaneous conditions. This approach provides human-relevant data to support route-switch decisions and formulation adjustments prior to clinical implementation.
By characterizing ISR risk early, sponsors can better inform IV-to-SC transition strategies and reduce uncertainty during development.
What endpoints can be evaluated in the ISR Platform®?
Depending on study design, ISR assessment may include:
- Multiplex analysis of secreted inflammatory mediators (e.g., cytokines and chemokines)
- Bulk RNA sequencing to characterize gene expression changes associated with local immune activation
- Pathway enrichment and bioinformatics-driven interpretation of transcriptomic data
- Tissue-level evaluation to assess structural and cellular responses
- Analysis across multiple donors to capture inter-individual variability
This flexible, multi-layered approach enables human-relevant characterization of ISR biology and supports informed non-clinical decision-making.
How does ex vivo human skin compare to animal models for ISR testing?
As a New Approach Methodology (NAM), this approach enables direct evaluation of human-specific local immune activation following subcutaneous or intradermal administration. This can support more translational insight into ISR risk while reducing reliance on animal testing for local tolerability assessment.
By leveraging human biology early in development, sponsors can strengthen the scientific rationale supporting formulation, route, and dosing decisions before clinical evaluation.