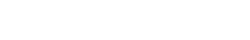Before proceeding to clinical trials in humans, today’s scientists study drug efficacy and toxicity in animals. Even though animal studies clearly raise ethical questions, it is considered even more unethical to conduct studies directly on humans without prior testing. Genoskin provides a unique alternative to animal testing and to human trials, using real, immunocompetent human skin.
Why human skin is a relevant study tool for drug and vaccine development
Human skin contains more than just structural cells
The skin is the body’s largest organ and offers the first protection barrier against external aggression, such as pollution, sunlight, chemicals, pathogens…. However, human skin is much more than an excellent tool to study the response of structural cells, such as melanocytes, keratinocytes, sebaceous glands, etc., to dermatological compounds and cosmetics. Real human skin also possesses an extraordinary diversity of immune cells that are involved in drug-induced immunotoxicity, including the most abundant concentration of resident memory T cells. (Tokura et al., 2021 & Kabashima et al., 2019)

A unique ecosystem that reflects the body’s immunological response
The immune cells we find in the skin (resident memory CD4 and CD8 T lymphocytes, dendritic cell subsets, innate lymphoid cells, natural killer cells, macrophages, mast cells, some B-cells and others… ) are also present in other human organs (Nguyen et al., 2019). In real skin, immune and structural cells constantly interact together to form a true ecosystem. Preserving such immune-structural interaction is key to apprehending immunological response to drugs prior to clinical trials.


Real, bio-stabilized and immunocompetent skin for relevant data
First-in-human data without harming humans or animals
Genoskin’s ex vivo human skin models are the only study platforms on the market, apart from clinical trials, to analyze potential immunotoxicity in a real, living, functional and immunocompetent human organ, the human skin. Our approach allows collecting first-in-human data without animal experiments and, ahead of clinical trials.

Using human skin to predict immunotoxicity in the body
All new human pharmaceuticals must be evaluated for their potential for immunomodulatory properties: immuno-suppression, injection site reactions, allergies, inflammation, cytokine storms, etc. Testing immunotoxicity in human skin can be used to predict systemic immunotoxicity in other human organs and assess the response of the entire human body to specific drugs, vaccines, cosmetics, medical devices, fillers, etc. The human skin in our ex vivo platforms comes from donated human skin, which is kept in a living state after surgery, thanks to our unique technology. As the skin is alive and bio-stabilized, it remains immunocompetent and provides a unique study tool for the pharmaceutical, biotech and cosmetic industries as well as for academic research and medical devices. All skin samples are collected with the informed consent of the donor and in full respect of all applicable regulations and procedures.

Genoskin’s ex vivo human skin platforms allow for the immune profiling of drugs, vaccines and compounds that are administered via topical, systemical, intradermal and subcutaneous routes. We provide the only testing platform worldwide to test subcutaneously injected products outside of animals and humans, to not only predict immune response to drugs in the skin but also in other human organs that share a similar immune ecosystem. Our expertise includes unbiased data analysis via artificial intelligence, spatial biology and data comparison to real-world evidence and drug data for extra relevance.
Comments are closed.