Skin Models
10
Sep
WHITE PAPER – HypoSkin® & immunogenicity testing
Explore how HypoSkin®—a standardized, injectable, and immunocompetent human skin model—provides a translational, animal-free solution for assessing the immunogenicity of therapeutic compounds. From structural stability to real-time immune response profiling, this white paper delivers data-backed insights designed for pharma and biotech teams advancing injectable drug development.
20
May
APPLICATION NOTE – NativeSkin® & Pharma
Explore how NativeSkin® supports ethical, human-relevant, and translational research for pharma.
2
Apr
Advancing medical device development with ex vivo human skin
Advancing medical device development with ex vivo human skinMedical devices are evolving rapidly, becoming more sophisticated and diverse in their applications. From injectable drug delivery systems to wound dressings, wearable sensors, and microneedles, these innovations must undergo rigorous testing to ensure safety, functionality, and biocompatibility. However, as medical devices become more complex, so do the challenges associated with their preclinicalRead more
18
Mar
APPLICATION NOTE – NativeSkin® & Cosmetics
Explore how NativeSkin® supports human-relevant cosmetic efficacy and safety testing, helping you strengthen claims and de-risk formulations earlier.
13
Mar
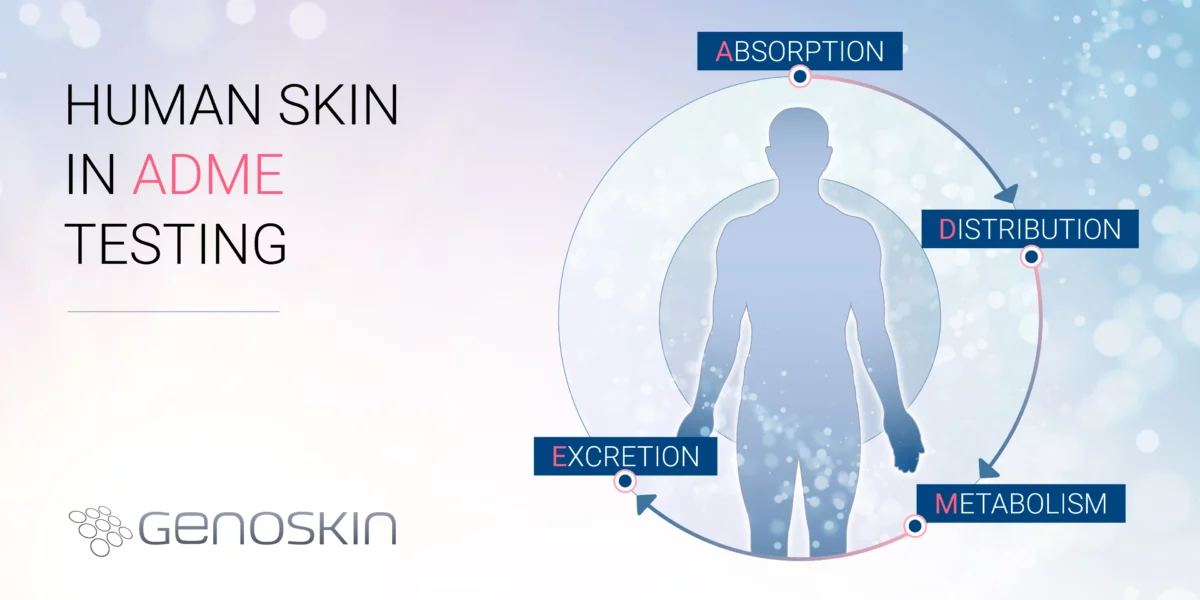
Beyond animal models: The role of human skin in ADME testing
Beyond animal models: The role of human skin in ADME testingADME (Absorption, Distribution, Metabolism, and Excretion) testing plays a vital role in modern drug development by improving candidate selection, reducing failures, enhancing safety assessments, ensuring regulatory compliance, and ultimately saving time and resources in bringing new drugs to market. Traditionally, ADME testing has relied on animal models and in vitroRead more
17
Feb
FACT SHEET – HairSkin, Human skin model with hair follicles
HairSkin®Human skin model with hair folliclesHairSkin®: the first ex vivo human skin model with mature pilosebaceous units HairSkin® is a cutting-edge ex vivo human skin model, developed with tissue containing fully mature pilosebaceous units. HairSkin® is designed to meet the requirements of both pharmaceutical and cosmetic industries and to empower innovation in dermatology, cosmetics, and personal care. Thanks to aRead more